HRCT is Cost-Effective in Screening for Diffuse Cystic Lung Diseases in Patients Presenting with a Spontaneous Pneumothorax
“A strategy of performing high-resolution computed tomography [HRCT] scanning followed by pleurodesis is cost-effective in screening for the presence of diffuse cystic lung diseases (DCLDs) in patients who present with an apparent primary spontaneous pneumothorax (PSP),” said Nishant Gupta, MD, assistant professor of medicine and director of the Interstitial Lung Diseases Center, University of Cincinnati (UC) Medical Center.
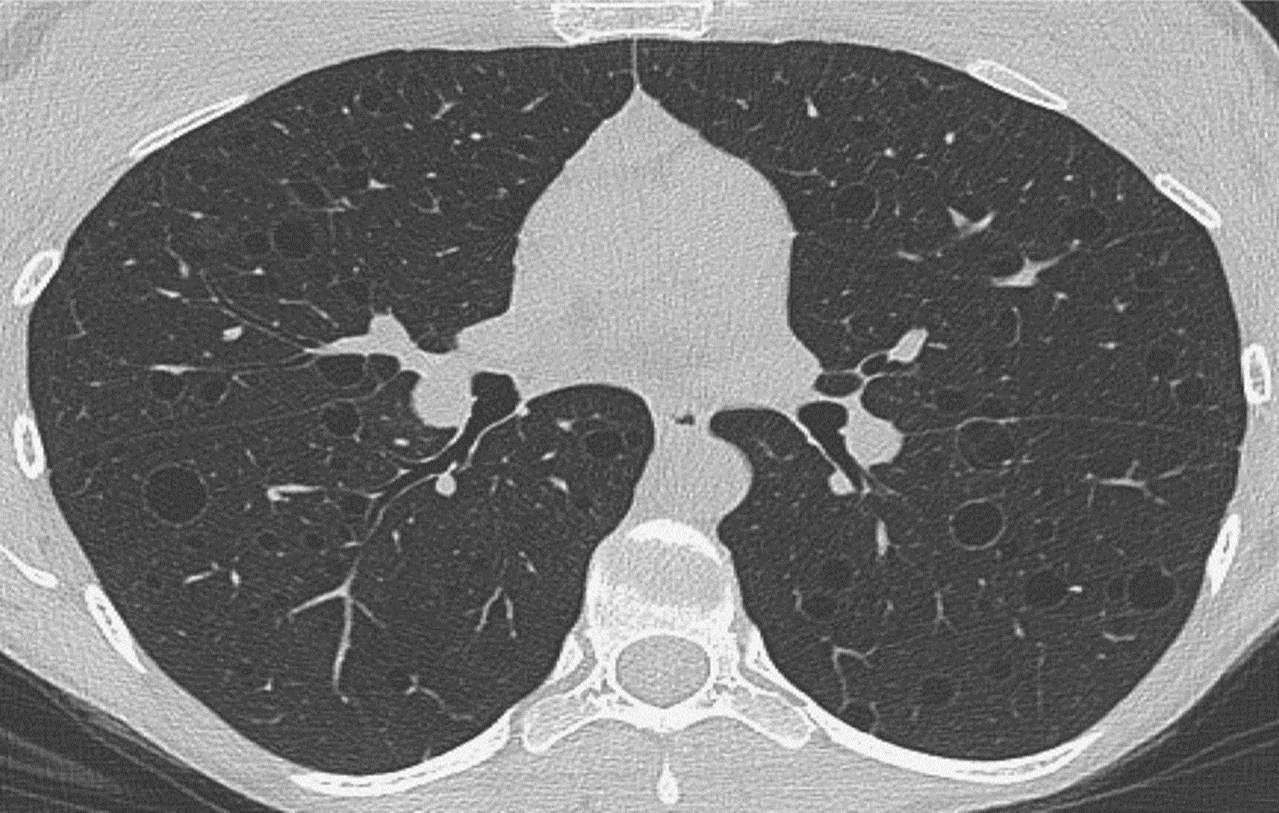
Figure 1: HRCT scan in a patient with LAM showing the characteristic thin-walled, round, uniform cysts present bilaterally in a diffuse distribution.
Dr. Gupta and his UC Medical Center co-authors reached this conclusion in a 2017 study that assessed the cost-effectiveness of HRCT chest imaging in the early diagnosis of Birt-Hogg-Dubé syndrome (BHD), lymphangioleiomyomatosis (LAM), and pulmonary Langerhans cell histiocytosis (PLCH)1.
According to the 2001 American College of Chest Physicians guidelines2, routine CT scans to evaluate for the presence of an underlying lung disease are not recommended in patients presenting with an apparent PSP. However, spontaneous pneumothorax is a common mode of presentation among patients with DCLDs, and “DCLDs such as BHD, LAM, and PLCH can be the underlying etiology of the pneumothorax in approximately 8-10% of the patients presenting with an apparent PSP,” Dr. Gupta explained1.
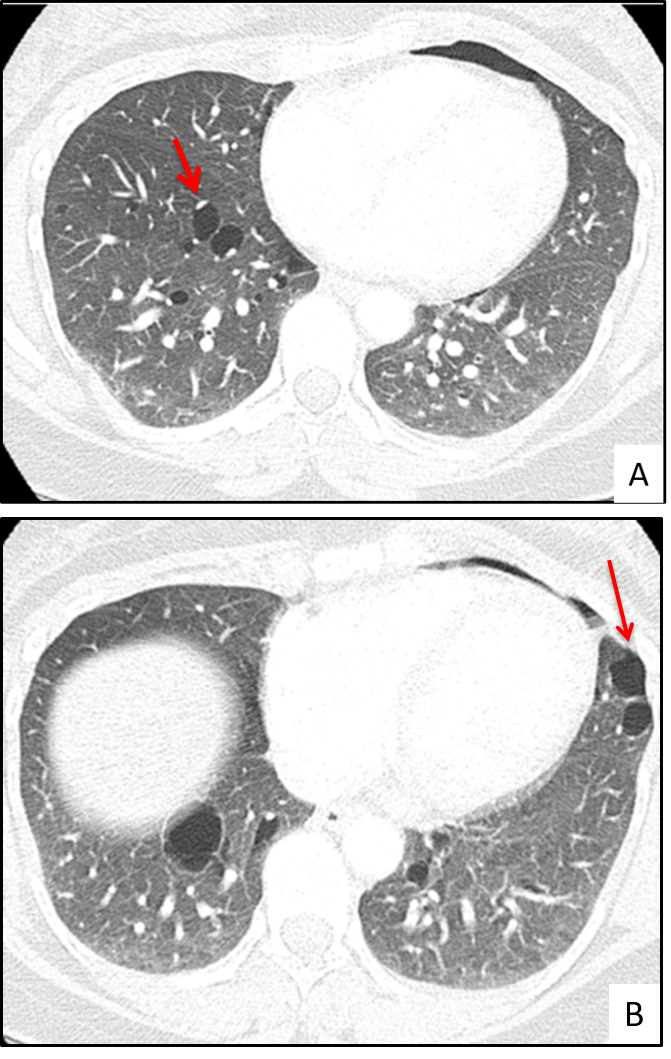
Figure 2: HRCT scans in two patients with BHD showing the characteristic oval/lentiform cysts present in a lower lobe distribution. Notice the predilection for pulmonary vasculature (2A, arrow), and the pleural surface (2B, arrow).
HRCT has significantly improved the ability of clinicians to diagnose these three diseases.3 A critical review of HRCT can reveal cyst characteristics that are unique to BHD, LAM and PLCH. For example, the cysts in LAM are thin-walled, round, uniform, and present diffusely in both lungs (Figure 1). In contrast, the cysts in PLCH tend to be more irregular in shape, can have thin as well thick walls, may have associated nodules, and are present in an upper-lobe predominant distribution (Figure 2). In patients with BHD, the pulmonary cysts are typically less numerous than LAM, tend to be more oval/lentiform in shape, are present mainly in the lower lobes, and have a strong predilection for the subpleural areas and the pulmonary vasculature (Figure 3)4,5.
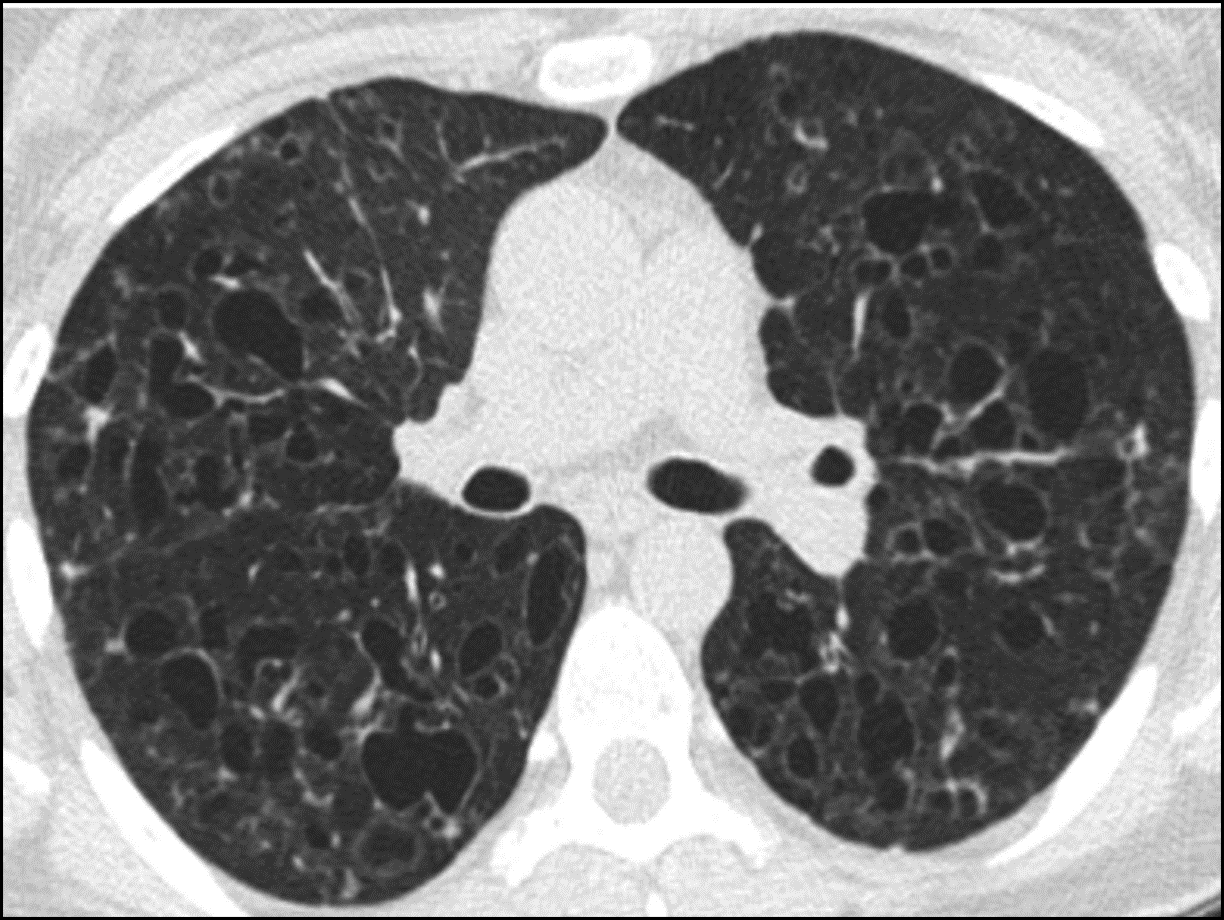
Figure 3: HRCT scan in a patient with PLCH showing the characteristic irregular/bizarre shaped cysts.
Early detection followed by timely pleurodesis (following the first episode of pneumothorax as opposed to waiting for a recurrent pneumothorax), said Dr. Gupta, would result in fewer pneumothorax recurrences. In patients with BHD, LAM and PLCH, the risk of recurrence after a first pneumothorax is 58%, 73% and 75%, respectively, as compared to a recurrence rate of approximately 30% in patients with PSP6-9. Dr. Gupta noted other disease-specific advantages to early detection. Because an effective, FDA approved treatment exists for patients with LAM10, early diagnosis could lead to drug intervention and prevent continued loss of lung function. BHD patients have a greater risk of renal cancer; earlier diagnosis would enable patients to enroll in surveillance programs for kidney tumors, as well as enable testing of asymptomatic family members for the presence of BHD5. In PLCH, which has a strong causal association with cigarette smoking, early diagnostic determination would facilitate counseling about smoking cessation, as well as provide an opportunity to screen for the presence of underlying genetic mutations.11
Dr. Gupta and his co-authors determined that using HRCT screening for LAM, BHD and PLCH followed by pleurodesis in patients presenting with an apparent PSP was both more costly and more effective. The marginal cost-effectiveness ratio of HRCT screening was $1,427 per quality-adjusted life-year (QALY) gained. Traditionally, strategies and interventions costing less than $50,000/QALY are characterized as extremely cost-effective. Based on these findings, the authors recommend “adult patients of all ages with an apparent PSP should undergo a screening chest HRCT scan to evaluate for the presence of underlying BHD, LAM, and PLCH.” 1
References:
1. Gupta N, Langenderfer D, McCormack FX, Schauer DP, Eckman MH. Chest Computed Tomographic Image Screening for Cystic Lung Diseases in Patients with Spontaneous Pneumothorax Is Cost Effective. Ann Am Thorac Soc 2017;14:17-25.
2. Baumann MH, Strange C, Heffner JE, et al. Management of spontaneous pneumothorax: an American College of Chest Physicians Delphi consensus statement. Chest 2001;119:590-602.
3. Gupta N, Meraj R, Tanase D, et al. Accuracy of chest high-resolution computed tomography in diagnosing diffuse cystic lung diseases. Eur Respir J 2015;46:1196-9.
4. Gupta N, Vassallo R, Wikenheiser-Brokamp KA, McCormack FX. Diffuse Cystic Lung Disease. Part I. Am J Respir Crit Care Med 2015;191:1354-66.
5. Gupta N, Sunwoo BY, Kotloff RM. Birt-Hogg-Dube Syndrome. Clin Chest Med 2016;37:475-86.
6. Almoosa KF, Ryu JH, Mendez J, et al. Management of pneumothorax in lymphangioleiomyomatosis: effects on recurrence and lung transplantation complications. Chest 2006;129:1274-81.
7. Mendez JL, Nadrous HF, Vassallo R, Decker PA, Ryu JH. Pneumothorax in pulmonary Langerhans cell histiocytosis. Chest 2004;125:1028-32.
8. Toro JR, Pautler SE, Stewart L, et al. Lung cysts, spontaneous pneumothorax, and genetic associations in 89 families with Birt-Hogg-Dube syndrome. Am J Respir Crit Care Med 2007;175:1044-53.
9. O’Rourke JP, Yee ES. Civilian spontaneous pneumothorax. Treatment options and long-term results. Chest 1989;96:1302-6.
10. McCormack FX, Inoue Y, Moss J, et al. Efficacy and safety of sirolimus in lymphangioleiomyomatosis. N Engl J Med 2011;364:1595-606.
11. Mourah S, How-Kit A, Meignin V, et al. Recurrent NRAS mutations in pulmonary Langerhans cell histiocytosis. Eur Respir J 2016;47:1785-96.
 Nishant Gupta, MD, MS
Nishant Gupta, MD, MS
Assistant Professor of Medicine
Director – Interstitial Lung Diseases Center,
Division of Pulmonary Critical Care and Sleep Medicine
University of Cincinnati Medical Center
PHONE: 513-558-4831
EMAIL: guptans@ucmail.uc.edu
Connect with Dr. Gupta on Doximity
Leave a reply →
