Bronchial Thermoplasty: A Promising Treatment for Patients with Severe, Refractory Asthma
Most patients with asthma are managed with a stepwise protocol in which therapy is escalated as disease severity worsens.1 Two recent additions to the asthma armamentarium, omalizumab and mepolizumab, are monoclonal antibodies to IgE and IL-5, respectively, that are aimed at asthma that remains inadequately controlled despite Global Initiative for Asthma (GINA) 2002 step 4 therapy. In two separate studies that included 577 patients, investigators found that omalizumab resulted in a significant reduction in the rate of clinically significant exacerbations, severe exacerbations, and emergency visits.2,3 Mepolizumab, the newest FDA-approved agent, was shown in the Dose Ranging Efficacy and Safety with Mepolizumab (DREAM) study of 158 patients to result in a relative reduction in the exacerbation rate of 47% in the intravenous-mepolizumab group and 53% in the subcutaneous-mepolizumab group. In addition, drug therapy resulted in significant reductions in composite outcome of emergency visits and hospitalizations, by 32% in the intravenous-mepolizumab group and 61% in the subcutaneous-mepolizumab group.4

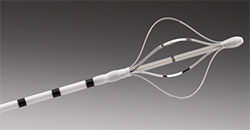
The Alair system is composed of two components. The Alair catheter is a flexible tube with an expandable wire array at the tip. It is introduced into the lungs by way of the working channel of a standard bronchoscope.
A New Agent: Bronchial Thermoplasty
For the 5% to 10% of asthma patients5 with uncontrolled symptoms despite all appropriate pharmacologic therapies, bronchial thermoplasty (BT) may improve quality of life by reducing bronchoconstriction and frequency of asthma exacerbations.6-8
The BT system consists of a flexible catheter with an expandable wire basket-like device at the tip that is introduced into the lungs through a standard bronchoscope. According to Peter Lenz, MD, MEd, assistant professor of medicine and program director for the pulmonary and critical care fellowship at the University of Cincinnati (UC) Medical Center, the controlled radiofrequency energy is transmitted through the basket tip to the airway wall and reduces airway smooth muscle mass.6-8 The procedure is performed in three treatment sessions a few weeks apart. In some patients, the benefit is remarkable. “One of my patients was able to taper off long-term prednisone, reduce inhaler use, and drop her annual hospitalization requirement from five stays to none,” said Dr. Lenz. “In some cases, BT may precipitate exacerbations of asthma and trigger a short inpatient stay or the need for short course prednisone.”
BT has been studied in three randomized controlled trials, including two that have collected data for up to five years.6-8 The Asthma Intervention Research (AIR) trial of 297 randomized patients with moderate to severe asthma demonstrated that BT was safe, supported by an absence of clinical complications (according to reporting of
The Alair radiofrequency controller supplies monopolar RF energy to the catheter. The heat is delivered to the airway wall at a precisely controlled at 65°C for 10 seconds. The radiofrequency controller contains multiple sophisticated safety algorithms to ensure safety. Images provided courtesy of Boston Scientific. © 2016 Boston Scientific Corporation or its affiliates. All rights reserved.
adverse events) and stable lung function, for five years after BT.6 In the AIR2 trial, the first blinded, sham treatment controlled study that involved 288 people with severe asthma from 30 centers, 79% of patients in the BT group and 64% patients in the sham group experienced clinically meaningful improvement in their asthma quality of life score. The BT group experienced a 32% reduction in severe exacerbations, an 84% reduction in emergency room visits for respiratory symptoms, and 66% fewer days lost from work/school/other daily activities because of asthma.7 The Research in Severe Asthma trial, which was a randomized, controlled clinical trial to evaluate the safety of BT in patients with severe and persistent asthma, demonstrated the five-year safety of BT in 14 patients who took part in the long-term follow-up.8 More BT trials are underway, and the University of Cincinnati Medical Center will be participating in two upcoming BT trials.
How Bronchial Thermoplasty works to reduce severe exacerbations. Video provided courtesy of Boston Scientific Corporation.
Candidates for Bronchial Thermoplasty
Who are candidates for BT? Sadia Benzaquen, MD, assistant professor and director of interventional pulmonology at UC Medical Center, said, “Patients with markedly impaired quality of life due to persistent asthma that has proven to be refractory to routine and advanced asthma interventions should be referred to the interventional pulmonary service (IPS) for evaluation.” Dr. Benzaquen attributes good outcomes from BT performed at the UC Medical Center to rigorous patient selection. Up to 20% of patients referred to UC IPS have been found to have respiratory conditions other than asthma, such as excessive dynamic airway collapse (EDAC) or vocal cord dysfunction. “I make no assumptions and evaluate the patient anew,” he said, “to rule out asthma mimics or treatable conditions that can make asthma worse, such as esophageal reflux.”
Success Rate and Outcomes
Another reason that BT is not performed more often, according to Dr. Benzaquen, is that health insurance approval is often very difficult to obtain in the absence of official guidelines for BT candidate selection. His team at UC Medical Center, he said, has a very good success rate in obtaining approval, and he has shared his BT-candidate protocol with many other institutions.
Dr. Benzaquen has performed the BT procedure on dozens of patients, and is obviously proud of the outcomes that his patients have enjoyed. In one example he shared: “A young man who was so severely asthmatic that he had been hospitalized almost weekly has not been hospitalized in three years, and he has had a significant reduction in the medications he takes.” “I am enthusiastic about the potential of this procedure to alleviate suffering in the small subset of asthma patients that have failed all other interventions.” Dr. Lenz and Dr. Benzaquen both stressed that BT is not curative, and that most patients continue to take asthma medications following the procedure, albeit often at lower doses.
References:
1. Pocket Guide for Asthma Management and Prevention. Global Initiative for Asthma. Updated 2015. Available at: http://ginasthma.org. Accessed: March 5, 2016.
2. Humbert M, Beasley R, Ayres J, et al. Benefits of omalizumab as add-on therapy in patients with severe persistent asthma who are inadequately controlled despite best available therapy. (GINA 2002 step 4 treatment): INNOVATE. Allergy. 2005;60(3):309-316.
3. Brussette G, Michils A, Louis R, et al. “Real-life” effectiveness of omalizumab in patients with severe persistent allergic asthma: The PERSIST study. Respir Med. 2009;103:1633-1642.
4. Ortega HG, Liu MC, Pavord DM, et al, for the MENSA Investigators. Mepolizumab treatment in patients with severe eosinophilic asthma. N Engl J Med. 2014;371:1198-1207.
5. The prevalence of severe refractory asthma. American Academy of Allergy Asthma and Immunology. Available at: http://www.aaaai.org/global/latest-research-summaries/New-Research-from-JACI-In-Practice/refractory-asthma.aspx. Accessed: March 5, 2016.
6. Thomson NC, Rubin AS, Niven RM, et al, for the AIR Trial Study Group. Long-term (5 year) safety of bronchial thermopolasty: Asthma Intervention Research (AIR) trial. BMC Pul Med. 2011;11:8. http://www.ncbi.nlm.nih.gov/pubmed/21314924.
7. Castro M, Rubin AS, Laviolette M, et al, for the AIR2 Trial Study Group. Asthma Intervention Research 2 (AIR2) Trial. Am J Respir Crit Care Med. 2010;181:116-124.
8. Pavord ID, Cox G, Thomson NC, et al, for the RISA Trial Study Group. Research in Severe Asthma (RISA) trial—5 year results. Safety of bronchial thermoplasty in patients with severe refractory asthma. Ann Allergy Asthma Immunol. 2013;111:402-407.
 Sadia Benzaquen, MD
Sadia Benzaquen, MD
Assistant Professor, Department of Internal Medicine
Director of Interventional Pulmonology
University of Cincinnati Medical Center
(513) 475-8523
benzaqsa@ucmail.uc.edu
Connect with Sadia Benzaquen, MD on Doximity
 Peter Lenz, MD, MEd, FCCP
Peter Lenz, MD, MEd, FCCP
Assistant Professor of Medicine
Fellowship Director for Pulmonary & Critical Care Medicine
University of Cincinnati
(513) 558-3916
lenzpr@ucmail.uc.edu
Connect with Peter Lenz, MD on Doximity
Leave a reply →
