Lymphangioleiomyomatosis Therapy Submitted for FDA Review
Lymphangioleiomyomatosis (LAM) is a rare, metastatic neoplasm of women that leads to cystic lung destruction and declining lung function.1 Until recently, LAM patients faced respiratory disability and the need for supplemental oxygen and lung transplantation without an effective therapy. However, research spearheaded by investigators at the University of Cincinnati Medical Center Division of Pulmonary, Critical Care & Sleep Medicine and Cincinnati Children’s Hospital Medical Center has offered clinicians a new approach to their patients with LAM. The drug sirolimus (also known as rapamycin) was shown to be effective in the treatment of LAM in the NIH- and FDA-funded Multicenter International Lymphangioleiomyomatosis Efficacy and Safety of Sirolimus (MILES) trial2, and is now being considered for FDA approval on the basis of that study. Although sirolimus is not curative, the drug stabilizes lung function and improves functional performance, and has been shown to be generally well-tolerated by patients with LAM. MILES demonstrated that randomized trials are feasible in even the rarest of lung diseases.
Uncommon lung diseases including LAM, pulmonary alveolar proteinosis, pulmonary Langerhan’s Cell Histiocytosis and Hermansky-Pudlak Syndrome are a focus for Division research, and in the Translational Pulmonary Science Center, a collaborative program with Cincinnati Children’s Hospital Medical Center. Frank McCormack, MD, Division of Pulmonary Director at the University of Cincinnati Medical Center, states of the current research climate, “The advances in rare lung disease science have been truly spectacular. The ability to approach disease pathogenesis from the vantage point of a primary molecular defect has been the catalyst that has taken the rare lung diseases from obscurity to therapies in our lifetimes.”
The Institute of Medicine of the National Academies of Science cited the MILES trial as an exemplar of successful translational research in its report on the state of Clinical and Translational Science Awards (CTSAs), and the National Institutes of Health (NIH) just awarded Cincinnati Children’s Hospital Medical Center, Vanderbilt Medical Center and University of Cincinnati Medical Center, Division of Pulmonary, Critical Care & Sleep Medicine a multi-million dollar grant to continue their rare lung disease studies. One of the supported projects will address the long-term risks and benefits of sirolimus, including assessments of tolerability and maintenance of efficacy, as well as the development of resistance.
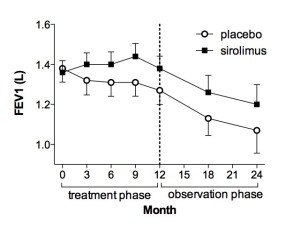
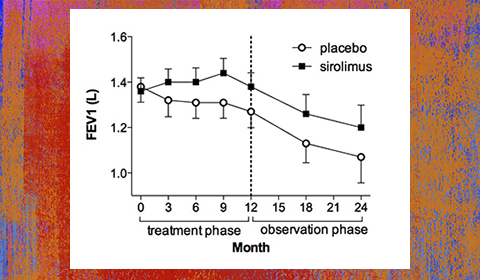
The MILES Trial. There were 89 patients randomized to placebo or sirolimus groups for one year, followed by one year of observation. Lung function (forced expiratory volume in one second-FEV1) stabilized on sirolimus relative to the placebo group, but declined again when the drug was held, suggest continuous therapy is required.
The MILES trial was funded by the Office of Rare Diseases, the National Center for Research Resources and the Food and Drug Administration, as well as 10 other sources. The LAM Foundation, a Cincinnati-based patient advocacy organization, contributed $500,000, assisted with travel and visit payments to sites and helped to recruit patients for the trial that was headquartered at the University of Cincinnati Medical Center and Cincinnati Children’s Hospital Medical Center. Patients were randomized to receive either placebo or drug for one year, and were observed off therapy for an additional year. The primary endpoint for the study was lung function. Patients who received placebo lost 10% of their lung function over the course of a year, but patients who took the drug had no loss of lung function during that same period. When the drug was withdrawn, lung function declined again, suggesting continued benefit requires continuous use.
MILES was an investigator-initiated trial, and was conducted without industry sponsorship. “This study was fueled by courageous patients and the altruism of physician scientists at 13 sites from Osaka to Portland.” McCormack elaborates: “The University of Cincinnati and Cincinnati Children’s also deserve a lot of praise for the generous manner in which they supported MILES. They waived their ‘indirects’ to preserve funds for the trial. They provided us with the flexibility to spend large amounts of time planning and executing the study. They funded the first grants that helped get us off the ground. Talented people emerged from all over the Academic Health Center at key points throughout the trial to help us reach a successful conclusion.”
In a recent LAM Foundation poll, it was estimated that over 30% of the US LAM population is now taking sirolimus. The drug was recently approved in Japan based on the results of MILES and an ongoing study (MLSTS) there. “There is limited accessibility in many parts of the world, which is precisely why FDA approval is so important,” says McCormack. “Approvals in other nations will very likely follow.”3
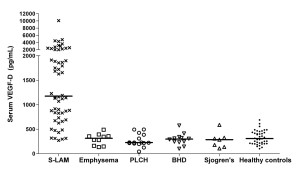
A VEGF-D level of 800 pg/ml or greater in a patient with typical thick walled cystic change distinguishes LAM from other mimics.5 The VEGF-D test is available commercially at https://research.cchmc.org/translationalcores/ttdsl. Sporadic LAM (S-LAM), pulmonary Langerhans’ cell histiocytosis (PLCH), Birt Hogg Dubé (BHD).
Other LAM research has been productive as well. A new ELISA blood test developed by UC Medical Center investigators assign the diagnosis of LAM without the need for surgical lung biopsy in a substantial fraction of patients.4,5 It is also useful prognostically.6 If a patient has an elevated level of this biomarker, lung function decline is typically more rapid. The good news is that these patients are also more likely to respond to therapy. The test is now commercially available on the campus of the University of Cincinnati Academic Health Center.7
The goal of UC Medical Center pulmonary research is to conduct studies that have the potential to favorably impact human health. The identification of a treatment for LAM in the MILES trial and the development of Vascular endothelial growth factor D (VEGF-D) (Figure 2) as a useful biomarker exemplify the translational research focus of the division, and the commitment to favorable outcomes for patients.

