UC Medical Center Researchers Advancing Precision Medicine
Researchers at the University of Cincinnati (UC) Medical Center are advancing the treatment of hypertrophic cardiomyopathy (HCM) and heart failure through the study and application of precision medicine (PM). “From the perspective of PM, a key goal involves uncovering the direct clinical relationship between genetic variants and disease phenotypes to allow for customized healthcare delivery and treatments tailored to the individual patient,” says Sakthivel Sadayappan, PhD, MBA, professor, department of internal medicine; division of cardiovascular health and disease; director of Heart Branch of the UC Heart, Lung and Vascular Institute, UC Medical Center.
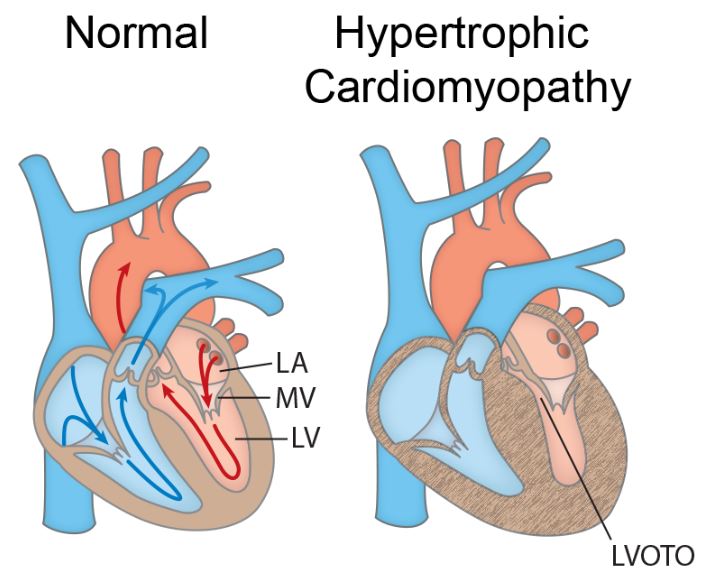
Anatomical cross sections of a normal heart and a heart with HCM shows generalized hypertrophy and reduced size of the LV chamber. Increased wall thickness of the interventricular septum and protrusion of the MV into the outflow path contribute to left ventricular outflow tract obstruction (LVOTO). The advancement of PM may give physicians more personalized treatment plans for patients. Image credit: Kraker J, Viswanathan SK, Knöll R and Sadayappan S (2016). Recent Advances in the Molecular Genetics of Familial Hypertrophic Cardiomyopathy in South Asian Descendants. Front. Physiol.7:499. doi:10.3389/fphys.2016.00499 licensed under CC BY 4.0.
HCM affects up to 500,000 people in the United States, is the most common form of cardiomyopathy and is the leading cause of sudden cardiac death in young athletes1. Currently, no therapies can completely cure HCM, says Dr. Sadayappan. Many current treatment regimens, including beta-blockers, calcium-channel blockers, or disopyramide, implantable cardioverter defibrillators (ICDSs), alcohol septal ablation and septal myectomy can have positive outcomes in symptomatic patients.
Despite advancements in treatment over the past 25 years, HCM remains a major cause of sudden cardiac death. Dr. Sadayappan believes that PM can help to determine why there is clinical variability in individuals with the same mutations. “We have learned that genetic defects can present variable disease outcomes, even within the same family. For example, some gene mutations result in increased left ventricular muscle mass, cardiac arrhythmias, or metabolic disorders. Therefore, no two HCM patients can be treated alike,” says Dr. Sadayappan.
On a PM platform, personal assessments should be performed before deciding on a treatment plan, says Dr. Sadayappan. Next-generation sequencing (NGS) of a patient’s entire genome can establish the presence or absence of secondary gene defects. Genetic data is useful to predict certain disease outcomes, given their variability. Moreover, siblings and children of patients could be tested for the presence of the same genetic defect as an important step toward predicting potential disease in positive carriers. In such cases, preventive approaches, such as changes in lifestyle, including diet and exercise, along with medications, could be taken.
Given the nature of HCM and its genetic complexity, future studies are expected to develop more pathways toward the delivery of PM for HCM, says Dr. Sadayappan. The American Heart Association and National Institutes of Health have both taken several critical steps toward individualizing diagnosis and treatment. Also, establishing gene correction of HCM mutations, either by CRISPR or gene therapy, before or after birth, is currently underway in several laboratories, including the laboratory at UC Medical Center. Several studies have already confirmed the feasibility of in vitro conditions and animal models.
Reference:
1. American Heart Association. Hypertrophic Cardiomyopathy. http://www.heart.org/idc/groups/heart-public/@wcm/@hcm/documents/downloadable/ucm_312225.pdf. Accessed 11 Dec 2017.
 Sakthivel Sadayappan, PhD, MBA
Sakthivel Sadayappan, PhD, MBA
Professor, Department of Internal Medicine
Director of Heart Branch of the UC Heart, Lung and Vascular Institute
University of Cincinnati Medical Center Division of Cardiovascular Health and Disease
Phone: (513) 558-7498
Email: sadayasl@uc.edu

